Table of Contents
Aflatoxicosis in Cattle and Buffaloes: The Hidden Threat Devastating Livestock Health and Farm Economics
Introduction: Understanding the Silent Killer in Your Livestock Feed
In the world of livestock farming, few threats are as insidious and economically devastating as aflatoxicosis. This mycotoxic disease, caused by toxic compounds produced by certain molds, represents one of the most significant challenges facing cattle and buffalo producers worldwide. Also known as “Degnala” disease in cattle and buffaloes, aflatoxicosis silently infiltrates herds through contaminated feed, causing widespread health problems, dramatic reductions in milk production, and substantial economic losses that can cripple farming operations.
The disease is caused by toxins produced by mold (fungus) that grows on feed such as groundnut cake, maize, and other stored fodder that becomes damp during storage.
What makes aflatoxicosis particularly dangerous is its ability to cause major losses in both health and performance of animals, with the economic impact of reduced productivity being many times greater than the impact caused by fatalities alone.
Milk production, a critical income source for dairy farmers, may drop by more than 15% in affected herds, representing a devastating blow to farm profitability.
What Are Aflatoxins and How Do They Form?
The Science Behind the Toxins
Aflatoxins are secondary metabolites produced by certain toxigenic fungi, primarily species of Aspergillus, particularly Aspergillus flavus and Aspergillus parasiticus
These microscopic organisms have the remarkable and unfortunate ability to produce potent hepatotoxins—compounds that are extremely toxic to the liver—when they grow on various agricultural commodities under favorable conditions.
The formation of aflatoxins is not a random occurrence but rather a complex biological process influenced by multiple environmental and agricultural factors. These toxins are low-molecular-weight compounds that can contaminate foods at all stages of production, from before and after harvest through processing, transport, and, critically, storage of both plants and grains.
This widespread potential for contamination makes aflatoxins a persistent threat throughout the entire agricultural supply chain.
Environmental Conditions That Promote Aflatoxin Production
Understanding the conditions that favor aflatoxin production is crucial for prevention. The fungi responsible for producing these toxins thrive under specific environmental conditions:
Temperature and Humidity: Hot temperatures combined with high humidity create the perfect storm for fungal growth and aflatoxin production. This is why aflatoxicosis is particularly prevalent in warmer climatic zones and during certain seasons when these conditions align
Moisture Content: The moisture content of stored feed is perhaps the most critical factor. When feed materials like rice straw, maize, or groundnut cake become damp—whether from improper storage, exposure to rain, or storage in humid conditions—the water activity increases to levels that support fungal proliferation
Storage Conditions: Poor on-farm storage of corn and other feed ingredients is a major contributor to aflatoxicosis outbreaks
Feed stored in lowlands near flooded areas, or straw stored soon after harvest without proper drying, provides ideal conditions for fungal development.
Stress Factors: Interestingly, aflatoxin production is often triggered by stress conditions affecting either the fungus or its plant host. Drought stress in crops, insect damage, and mechanical injury to plants can all increase susceptibility to fungal colonization and subsequent toxin production.
Common Feed Sources of Contamination
Aflatoxins can contaminate a wide variety of feedstuffs, but certain materials are particularly susceptible:
Cereal Grains: Maize (corn) is perhaps the most commonly contaminated feed ingredient, widely used as an energy source in animal feed and representing 50-80% of many animal diets
Wheat, sorghum, and rice are also susceptible.
Oilseed Meals: Oilseed meals contain a higher incidence of aflatoxin contamination (up to 96%) compared to cereals
Groundnut cake, cottonseed meal, and soybean meal are particularly vulnerable.
Rice Straw: In rice-producing regions, rice straw serves as the primary winter forage for ruminants and is strongly associated with Degnala disease
When improperly stored, rice straw becomes a major source of exposure to mycotoxins.
Other Agricultural By-products: Various crop residues, dried grasses, and hay can harbor toxigenic fungi if not properly dried and stored.
How Aflatoxicosis in Cattle and Buffaloes Affects Humans
The Food Chain Connection: From Livestock to Human Exposure
When cattle and buffaloes develop aflatoxicosis from consuming contaminated feed, the consequences extend far beyond the farm gate. Humans become exposed through multiple pathways in the food chain, creating significant public health concerns that affect consumers, farmers, and entire communities.
Primary Routes of Human Exposure
- Milk and Dairy Products: The Most Critical Pathway
The most documented route of human exposure is through milk contamination. When dairy animals consume feed containing aflatoxin B1 (AFB1), their livers metabolize it into aflatoxin M1 (AFM1), which is then excreted into milk
ResearchGate
This “carry-over” phenomenon means that even if animals show no clinical signs of illness, their milk can still contain hazardous levels of AFM1.
Key facts about milk transmission:
- AFM1 can be detected in milk within hours after an animal ingests contaminated feed
Cambridge University Press & Assessment
- Transfer rates from feed to milk typically range from 1-2%, though high-yielding cows (>30 kg milk/day) may show rates up to 6%
- AFM1 is heat-stable, meaning pasteurization, boiling, or standard dairy processing does not eliminate the toxin
- The toxin can also transfer into dairy products like cheese, yogurt, and butter, with varying concentrations depending on processing methods
ResearchGate
- Meat and Offal Consumption
While less studied than milk transmission, aflatoxins and their metabolites can accumulate in animal tissues:
- Liver and kidney tissues may contain higher concentrations due to the organs’ role in toxin metabolism
- Muscle tissue generally has lower levels but is not risk-free
- Animals slaughtered during acute aflatoxicosis episodes may have elevated tissue residues
- Indirect Exposure Through Environmental Contamination
- Manure from affected animals can contaminate soil and water sources
- Improper disposal of contaminated carcasses or feed can spread fungal spores
- Dust from moldy feed storage areas may carry spores that affect farm workers and nearby residents
Human Health Risks: From Acute Toxicity to Chronic Disease
Acute Aflatoxicosis in Humans
While rare, high-dose exposure to aflatoxins can cause acute poisoning with severe, sometimes fatal outcomes:
Symptoms of acute exposure
- Nausea, vomiting, and abdominal pain
- Jaundice (yellowing of skin and eyes)
- Acute liver injury with elevated liver enzymes
- Bleeding disorders due to impaired clotting factor synthesis
- Edema (fluid retention) and ascites
- Convulsions, lethargy, and in severe cases, coma or death
High-risk scenarios:
- Consumption of milk from severely affected herds during outbreak periods
- Use of contaminated animal products in emergency food aid without proper screening
- Occupational exposure among farmers handling moldy feed without protection
Chronic Health Effects: The Greater Public Health Burden
Far more common than acute poisoning is the insidious impact of long-term, low-level exposure:
- Liver Cancer (Hepatocellular Carcinoma)
Aflatoxin B1 is classified as a Group 1 human carcinogen by the International Agency for Research on Cancer (IARC)
Key facts:
- Chronic exposure is responsible for an estimated 4.6% to 28.2% of global liver cancer cases
- The risk is dramatically amplified in people infected with the hepatitis B virus (HBV); the combination of HBV and aflatoxin exposure increases liver cancer risk up to 60-fold compared to either factor alone
- The carcinogenic mechanism involves DNA damage: aflatoxin metabolites bind to DNA, causing characteristic mutations in the p53 tumor suppressor gene (specifically a G→T transversion at codon 249)
- Child Growth Impairment and Developmental Effects
Children are disproportionately affected by aflatoxin exposure:
- Multiple studies associate aflatoxin exposure with stunted growth in children, though confounding factors like malnutrition and infection complicate causal attribution
- Proposed mechanisms include:
- Impaired nutrient absorption due to intestinal damage
- Reduced appetite and feed efficiency
- Chronic inflammation and immune activation divert energy from growth
- Maternal exposure during pregnancy is linked to higher rates of preterm birth and late-term miscarriage
- Immune System Suppression
Aflatoxins compromise immune function through multiple pathways:
- Reduced production and function of key immune cells (T-cells, B-cells, macrophages)
- Impaired antibody responses to vaccines and infections
- Increased susceptibility to infectious diseases, including HIV progression and tuberculosis reactivation
- This immunosuppression creates a vicious cycle: malnourished, immunocompromised individuals are more vulnerable to infections that further impair growth and development
- Other Potential Health Effects
Emerging research suggests additional concerns:
- Genotoxicity: Aflatoxins can damage DNA in multiple cell types, potentially contributing to cancers beyond the liver
- Reproductive effects: Animal studies show reduced fertility and developmental abnormalities; human data are limited but concerning
- Neurological effects: Some evidence links chronic exposure to cognitive impairment, though mechanisms remain unclear
Vulnerable Populations: Who Is at Greatest Risk?
Infants and Young Children
Children face heightened vulnerability due to:
- Higher relative intake: Per kilogram of body weight, children consume more milk and food than adults
- Developing organs: Immature liver detoxification systems and rapidly dividing cells are more susceptible to toxin damage
- Critical growth windows: Exposure during key developmental periods can have lifelong consequences
- Breast milk transmission: AFM1 can appear in human breast milk when mothers consume contaminated dairy products
Pregnant and Lactating Women
- Toxins can cross the placental barrier, potentially affecting fetal development
- Lactating women consuming contaminated dairy may transfer AFM1 to infants via breast milk
- Physiological stresses of pregnancy may reduce detoxification capacity
Immunocompromised Individuals
People with HIV/AIDS, chronic hepatitis, malnutrition, or other conditions affecting immunity face:
- Reduced ability to detoxify and eliminate aflatoxins
- Greater susceptibility to opportunistic infections exacerbated by aflatoxin-induced immunosuppression
- Potentially accelerated disease progression
Rural and Low-Income Communities
Populations in aflatoxin-endemic regions face compounded risks:
- Limited access to diverse, high-quality food sources increases reliance on potentially contaminated staples
- Inadequate feed storage infrastructure perpetuates contamination cycles in livestock
- Limited healthcare access delays diagnosis and treatment of aflatoxin-related conditions
- Economic pressures may force consumption of visibly moldy but “salvageable” food
Regulatory Frameworks and Safety Standards
International Guidelines
Recognizing the public health threat, multiple agencies have established limits:
| Agency/Region | Limit for AFM1 in Milk | Notes |
| European Union | 0.05 µg/kg (ppb) | Among the strictest globally |
| United States (FDA) | 0.5 µg/kg (ppb) | Action level for interstate commerce |
| Codex Alimentarius | 0.5 µg/kg (ppb) | International reference standard |
| Many developing countries | 0.5–1.0 µg/kg | Varies by national capacity |
Challenges in Enforcement
Despite established standards, implementation faces obstacles:
- Testing capacity: Many regions lack laboratories equipped for sensitive mycotoxin analysis
- Informal markets: Small-scale dairy sales often bypass regulatory oversight
- Economic pressures: Farmers may resist discarding contaminated milk due to income loss
- Awareness gaps: Consumers and producers may not recognize the invisible nature of the risk
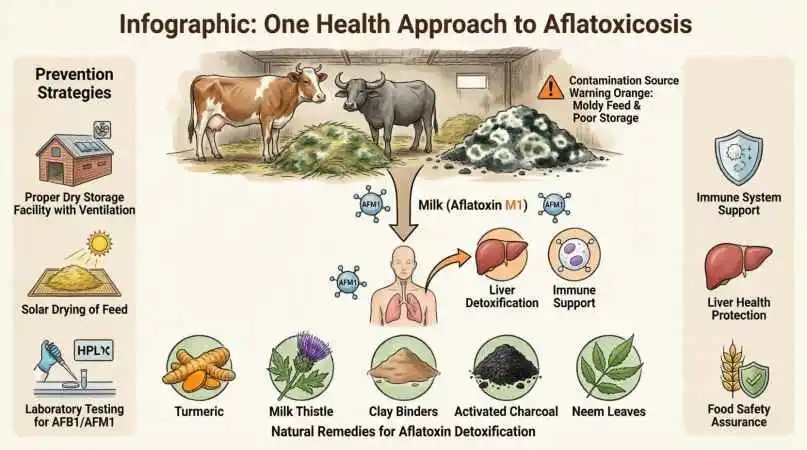
Risk Mitigation Strategies: Protecting Human Health
At the Farm Level: Breaking the Contamination Cycle
- Prevent Feed Contamination (Most Effective Strategy)
- Dry feed to ≤13% moisture before storage
- Store in dry, ventilated facilities away from flood zones
- Test high-risk feeds (maize, groundnut cake, rice straw) before use
- Remove and destroy visibly moldy feed immediately
-
Use Toxin Binders Strategically
- Aluminosilicate clays and yeast cell wall products can reduce AFM1 excretion in milk by 30-70% when added to feed.
- Important caveat: Binders reduce but do not eliminate risk; they complement, not replace, good feed management
-
Monitor Animal Health and Milk Quality
- Track milk production trends; sudden drops may signal toxin exposure
- Implement routine milk testing in high-risk regions
- Isolate and manage affected animals to prevent contaminated milk from entering the food supply
At the Processing Level: Reducing Consumer Exposure
- Milk Testing Programs
- Screen bulk tank milk for AFM1 using ELISA or HPLC methods
- Establish clear protocols for rejecting contaminated batches
- Provide feedback to farmers to improve on-farm practices
- Processing Considerations
- While heat treatment doesn’t destroy AFM1, some processing steps may reduce concentrations:
- Fermentation (yogurt, cheese) may slightly reduce levels through microbial activity
- Adsorbent materials added during processing show experimental promise but require regulatory approval
- Supply Chain Traceability
- Implement systems to trace contaminated milk back to source farms
- Use data to target extension services and support to high-risk producers
At the Consumer Level: Practical Protection Measures
- Diversify Food Sources
- Avoid relying exclusively on single-source dairy products
- Rotate brands and suppliers when possible
- Support Safe Production
- Purchase from producers with documented feed testing and quality control programs
- Advocate for stronger food safety regulations in your community
- Recognize Limitations of Home Remedies
- Boiling milk does NOT eliminate AFM1
- Visual inspection cannot detect mycotoxin contamination
- “Organic” or “natural” labels do not guarantee mycotoxin-free products
The Broader Public Health Perspective
Economic and Social Dimensions
Aflatoxin contamination creates cascading impacts:
- Trade barriers: Countries with high contamination levels face export restrictions, limiting economic development
- Food security paradox: Efforts to increase food production may inadvertently increase mycotoxin exposure if storage and quality control lag behind
- Healthcare costs: Treating aflatoxin-related illnesses diverts resources from other public health priorities
Climate Change Amplifies Risks
Emerging evidence suggests climate change may worsen aflatoxin problems:
- Warmer temperatures and altered rainfall patterns expand the geographic range of toxigenic fungi
- Increased frequency of drought and flood events stresses crops, making them more susceptible to fungal infection
- Longer growing seasons in some regions may allow multiple fungal infection cycles
One Health Approach: Integrating Animal, Human, and Environmental Health
Effective management requires coordination across sectors:
- Veterinary services: Monitor livestock health and feed quality
- Public health agencies: Track human exposure and disease outcomes
- Agricultural extension: Educate farmers on prevention practices
- Food safety authorities: Enforce standards and support testing infrastructure
- Research institutions: Develop improved detection methods, resistant crop varieties, and mitigation technologies
Key Takeaways for Stakeholders
For Farmers and Livestock Producers
Your feed management decisions directly impact consumer health
Investing in proper storage and testing protects both your animals and your market access.
Early intervention when contamination is suspected prevents larger outbreaks.
For Dairy Processors and Retailers
Implementing milk testing builds consumer trust and reduces liability
Supporting farmer education creates a more resilient supply chain.
Transparent communication about safety measures strengthens brand reputation.
For Public Health Officials
Surveillance systems should integrate animal and human health data
Community education should emphasize that aflatoxins are invisible but preventable.
Policy interventions should balance food security with food safety
For Consumers
Choose dairy products from reputable sources with quality assurance programs.
Understand that price alone doesn’t guarantee safety; ask about testing practices.
Advocate for stronger food safety standards in your community
Conclusion: A Shared Responsibility
Aflatoxicosis in cattle and buffaloes is not merely an animal health issue—it is a food safety and public health challenge with far-reaching consequences. The transmission of aflatoxin M1 through milk creates a direct pathway from contaminated feed to human exposure, with risks ranging from acute poisoning to chronic diseases like liver cancer.
The good news is that this threat is largely preventable. By implementing evidence-based practices at every stage—from feed storage on the farm to testing at the processing plant to informed choices by consumers—we can break the contamination cycle and protect both livestock productivity and human health.
As the search results emphasize: “Regulating aflatoxin levels in food and feed is essential for protecting public health and promoting equitable trade.”
This requires sustained investment, cross-sector collaboration, and a commitment to the One Health principle that animal welfare, food safety, and human health are inextricably linked.
The choice is clear: proactive prevention today prevents human suffering tomorrow. Every stakeholder in the food chain has a role to play in ensuring that the milk and meat reaching our tables are not just nutritious but also safe.
FAQS
- How does aflatoxicosis in cattle and buffaloes directly affect human health?
When livestock consume aflatoxin-contaminated feed, their livers convert aflatoxin B1 into aflatoxin M1, which is excreted in milk. Humans consuming this contaminated milk or dairy products face risks including liver damage, immune suppression, increased cancer risk (especially liver cancer), and impaired child development. The toxin is heat-stable, so pasteurization or boiling does not eliminate it.
- What natural remedies can help detoxify livestock affected by aflatoxicosis?
Several natural approaches show promise:
- Activated charcoal: Binds toxins in the gastrointestinal tract, reducing absorption (1-2% of body weight orally)
- Clay-based binders: Bentonite and zeolite clays can adsorb aflatoxins in the gut
- Turmeric (Curcuma longa): Contains curcumin with hepatoprotective and antioxidant properties
- Milk thistle (Silybum marianum): Silymarin supports liver regeneration and detoxification
- Neem (Azadirachta indica): Traditional use for immune support and antifungal properties
Note: Always consult a veterinarian before administering any remedy, as efficacy varies and proper dosing is critical.
- Can dietary changes in livestock reduce aflatoxin transmission to humans?
Yes. Strategic feeding practices can significantly reduce risk:
- Increase fiber intake: High-quality roughage dilutes toxin concentration and supports gut health
- Add antioxidant-rich feeds: Fresh green fodder, carrots, and pumpkin provide vitamins A, C, and E that support liver detoxification
- Include probiotic feeds: Fermented feeds or yogurt cultures may enhance rumen microbial capacity to degrade mycotoxins
- Balance protein sources: Adequate protein supports the synthesis of detoxification enzymes in the liver
- What lifestyle changes can farmers adopt to prevent aflatoxicosis outbreaks?
Feed Management Lifestyle Shifts:
- Establish a routine of inspecting stored feed weekly for mold, odor, or moisture
- Adopt “first-in, first-out” feed usage to minimize storage time
- Create a dedicated dry storage area with raised platforms and ventilation
- Keep detailed records of feed sources, test results, and animal health observations
Personal Protective Practices:
- Wear masks and gloves when handling suspect feed to reduce inhalation exposure
- Wash hands and change clothes after handling moldy materials before entering living areas
- Store animal feed separately from human food supplies
- Are there herbal supplements that support liver health in exposed animals?
Several herbs traditionally used in veterinary practice may support hepatic function:
- Silymarin (Milk Thistle extract): 10-20 mg/kg body weight; shown to protect liver cells from toxin damage
- Andrographis paniculata: Traditional use for liver support and immune modulation
- Phyllanthus niruri: Studied for hepatoprotective effects in animal models
- Guduchi (Tinospora cordifolia): Ayurvedic herb with antioxidant and immunomodulatory properties
Important: Herbal supplements should complement, not replace, removal of contaminated feed and veterinary care. Quality, dosage, and potential interactions require professional guidance.
- How can consumers protect themselves from aflatoxin exposure through dairy?
Practical Consumer Lifestyle Changes:
- Source diversification: Purchase milk from multiple reputable suppliers to avoid consistent exposure from a single contaminated source
- Choose certified products: Look for dairy brands that publish mycotoxin testing results or hold food safety certifications
- Support local transparent producers: Build relationships with farmers who practice open feed management and testing
- Limit high-risk products: During known outbreak seasons in your region, moderate consumption of products from high-risk supply chains
Home Practices:
- Store milk properly (refrigerated, sealed) to prevent secondary mold growth
- Avoid consuming dairy from animals showing signs of illness or reduced production
- When traveling to endemic regions, exercise caution with unpasteurized dairy products
- Can probiotics help reduce aflatoxin absorption in livestock?
Emerging research suggests certain probiotic strains may mitigate aflatoxin effects:
- Lactobacillus species: Some strains can bind aflatoxins in the gastrointestinal tract, reducing bioavailability
- Saccharomyces cerevisiae (brewer’s yeast): Yeast cell walls contain mannans that adsorb mycotoxins
- Bifidobacterium strains: May enhance gut barrier function and support immune response
Practical application: Incorporate probiotic supplements or fermented feeds (like properly prepared silage) as part of a comprehensive prevention strategy. Effects are strain-specific and dose-dependent.
- What natural feed additives can bind aflatoxins before they harm animals?
Evidence-Supported Natural Binders:
- Bentonite clay: 0.5-2% of diet; aluminosilicate structure adsorbs aflatoxins effectively
- Zeolite: Similar mechanism to bentonite; may also provide beneficial minerals
- Yeast cell wall products: Glucomannans in Saccharomyces cerevisiae bind multiple mycotoxin types
- Activated charcoal: Broad-spectrum adsorbent; use short-term during suspected exposure (can also bind nutrients)
Usage considerations: Binders work best as preventive measures or early interventions. They do not reverse existing tissue damage and should be combined with the removal of contaminated feed.
- How does stress management in livestock affect aflatoxin susceptibility?
Chronic stress compromises immune function and detoxification capacity, making animals more vulnerable to mycotoxin effects. Natural stress-reduction strategies include:
- Adequate space and ventilation: Reduces heat stress and respiratory challenges
- Consistent routines: Predictable feeding and handling schedules lower cortisol levels
- Social grouping: Maintain stable herd structures to minimize social stress
- Enrichment: Access to pasture or varied environments supports behavioral health
Reduced stress supports better feed conversion, stronger immunity, and more efficient toxin metabolism.
- Can dietary antioxidants in livestock feed reduce human health risks?
Yes, indirectly. Antioxidant-rich feeds support animal liver function, potentially reducing the amount of aflatoxin M1 excreted in milk:
- Vitamin E sources: Fresh green fodder, wheat germ, sunflower seeds
- Vitamin C sources: Amla (Indian gooseberry), citrus by-products (where palatable)
- Carotenoids: Carrots, pumpkin, marigold petals provide beta-carotene
- Polyphenols: Neem leaves, turmeric, green tea extracts (research ongoing)
Healthier animals with robust detoxification systems produce safer milk, creating a protective cascade from farm to consumer.
- What lifestyle changes help farmers detect aflatoxicosis early?
Daily Observation Routines:
- Monitor feed intake at each feeding; sudden drops warrant investigation
- Check milk production records daily; >10% unexplained decline triggers feed review
- Perform weekly visual exams of tails, hooves, and ears for early lesion signs
- Keep a “health journal” noting subtle changes in behavior or appearance
Environmental Awareness:
- Track local weather patterns; high humidity periods increase contamination risk
- Note seasonal feed transitions; winter straw feeding requires extra vigilance
- Participate in farmer networks to share outbreak alerts and prevention tips
- Are there traditional farming practices that naturally prevent feed contamination?
Many indigenous knowledge systems offer valuable prevention strategies:
- Solar drying traditions: Spreading grain or straw on raised mats in direct sunlight reduces moisture effectively
- Botanical storage protectants: Neem leaves, eucalyptus, or citronella placed in storage areas may deter fungal growth
- Ash or lime treatment: A light coating of wood ash on stored grains can reduce moisture absorption
- Elevated granaries: Traditional raised storage structures with thatched roofs promote ventilation
These practices, when combined with modern moisture monitoring, create robust, low-cost prevention systems.